Intramedullary Nail Series – Femoral Intertrochanteric Nail System: Deliver secure outcomes with ease, mastering procedures effortlessly.
Hip fractures refer to fractures occurring in the proximal femur and are one of the most common fractures throughout the body, accounting for approximately 6.73% of all fractures. Among these, intertrochanteric fractures are the most common manifestation of hip fractures, constituting about 3.4% of all bodily fractures. Particularly in the elderly, the incidence of intertrochanteric fractures is higher due to the presence of osteoporosis.

The Intramedullary Nail Series is a comprehensive intramedullary nail system developed by Decon Medical. Leveraging extensive clinical experience, precise anthropometric databases of the Chinese population, a dedicated biomechanical validation team, and robust manufacturing capabilities, it is designed to address fractures of long tubular bones in the extremities. This next-generation intramedullary nail system and corresponding instruments provide Chinese fracture patients with more suitable, innovative intramedullary solutions.
Specifically for intertrochanteric fractures of the hip, the Femoral Trochanteric Nail System within the series offers two intramedullary nail products tailored to the physiological structure of the Chinese femur, along with a streamlined set of surgical instruments compatible with both nail types. This system delivers smooth procedures and secure outcomes, truly enabling clinicians to "master the hip intervention with ease".
To validate the safety and effectiveness of the Trochanteric Femoral Nail System within the Intramedullary Nail Series, Atom Medical has continuously engaged in in-depth communication with professional clinicians and patients. Statistical analysis was conducted on adverse events associated with existing intramedullary nails for treating related conditions in the market, identifying clinical risks inherent to trochanteric femoral nails.

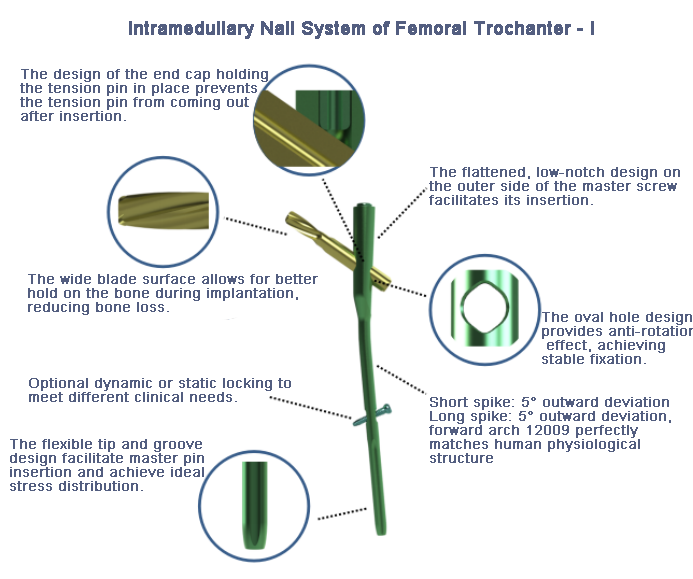
Research indicates that the most prevalent failure modes for femoral trochanteric intramedullary nails in clinical practice are main nail fracture or deformation (63%), lag screw cut-out or backing out (15%), and lag screw/locking screw fracture or deformation (9%). In response to these clinically reported issues, Atom Medical's specialized R&D team conducted an in-depth failure mode analysis. Corresponding test protocols were designed. Utilizing results from finite element analysis (FEA) and physical testing, the team iteratively performed "optimization, validation, and further refinement" on the product's most critically stressed locations during loading. Comparative analysis was also performed against femoral trochanteric intramedullary nail systems from leading domestic and international medical device companies. This process resulted in an excellent design where Dekang Medical's femoral trochanteric intramedullary nail system achieves superior performance compared to competing products.
It is well known that intramedullary nail instrumentation has always been a primary clinical focus. Atom Medical has consistently adhered to the principle of "products developed from clinical practice and dedicated to clinical service" in the development of its intramedullary nail series. During the design process of the intramedullary nail system, priority is given to the clinical application of the instrumentation. Key instruments, particularly the targeting devices, undergo repeated simulation and testing. Experience from predecessors is constantly summarized with continual refinement, aiming to design instrumentation that facilitates physician use and simplifies intramedullary nailing procedures.
Specifically for the femoral trochanteric intramedullary nail system, Atom Medical has systematically compiled and analyzed feedback regarding the application of these two types of femoral trochanteric nails in clinical practice. Through innovative development, the company has created a set of femoral trochanteric intramedullary nail instrumentation compatible with both types of nails. This achieves the goal of "a single set of instruments for two types of nails, addressing proximal femoral fractures."